AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Pacing failure to capture12/11/2023  In addition, undersensing may be due to altered cardiac signal morphology secondary to disease progression myocardial ischemia or infarction inflammatory changes or fibrosis at the lead-tissue interface, transient metabolic or electrolyte imbalance or the appearance of bundle-branch block or ectopy. If it occurs later, it could be due to battery depletion, system component failure, or functional undersensing (see below). Undersensing occurring shortly after implantation may be due to lead dislodgement or malposition or to cardiac perforation. 3,4As with failure to capture, the onset of undersensing relative to the time of device implantation helps identify the cause. Table 4elaborates on previously identified causes of undersensing. Therefore, the filtered signal may not be of sufficient size to be recognized as a valid event consequently, undersensing may occur. 3A signal with apparently adequate amplitude may be markedly attenuated by the sense amplifier if it has a reduced slew rate. The cardiac electrogram must have adequate amplitude and frequency content (slew rate) to be sensed properly. Drugs or imbalances that increase pacing thresholds ( table 2) may also increase latency. Finally, failure to capture may be misdiagnosed because of increased latency, which is the delay between stimulation and the onset of myocardial depolarization. 20Newer inhalation anesthetics, intravenous agents, narcotics, and anesthetic adjuncts have not been shown to affect thresholds. It is notable that addition of equipotent halothane, enflurane, or isoflurane to opiate-based anesthesia after cardiopulmonary bypass did not increase pacing thresholds. 3Transient, metabolic, and electrolyte imbalance, 6–12as well as drugs and other factors, 3,13–19may increase pacing thresholds ( table 2), a circumstance explaining pacing failure. 
Nonetheless, pacing thresholds may continue to rise until they exceed maximum pulse-generator output (exit block). As for causes ( table 1), stimulation thresholds may rise during lead maturation (2–6 weeks after implantation), but this has become far less of a problem since the introduction of steroid-eluting leads and other refinements in lead technology. 
3,4Event markers will identify the release of stimuli and recycling of the device by sensed events. To confirm this diagnosis, the device must be interrogated to examine event markers and measured data ( e.g. , lead impedances and pacing and sensing thresholds). With failure to capture, there will be visible pacing artifacts in the 12-lead surface electrocardiogram but no or intermittent atrial or ventricular depolarizations. Finally, the sense amplifier may detect isoelectric extrasystoles ( i.e. , in the surface electrocardiogram) that properly inhibit stimulus delivery. 
If so, the intervals between paced beats appear longer than normal. 5Failure to pace may be misdiagnosed with too-rapid strip-chart recording speeds. Examples of elective replacement indicators are listed in table 3. Misdiagnosis of failure to pace may occur with impending battery depletion, evidenced by the “elective replacement indicator.” The elective replacement indicator rate is not necessarily the same as the nominally programmed rate. When failure to pace occurs within 48 h of device implantation, lead dislodgement, migration, and myocardial perforation are probable causes. 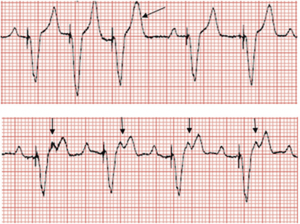
In addition, problems with the lead–tissue interface may explain failure to pace. Other causes are an open circuit caused by a broken, dislodged, or disconnected lead, lead insulation defects, or malfunction of other system components. 3,4įailure to pace is often due to oversensing (see Oversensing). 3,4To diagnose device malfunction, it is necessary obtain a 12-lead electrocardiogram and chest radiograph and to interrogate the device to check pacing and sensing thresholds, lead impedances, battery voltage, and magnet rate. Pacemaker malfunction is classified as failure to pace, failure to capture, pacing at abnormal rates, undersensing (failure to sense), oversensing, and malfunction unique to dual-chamber devices ( table 1). This encourages the emergence of intrinsic rhythm. With rate hysteresis, the pacing cycle duration is longer after a sensed versus paced depolarization. 3For example, failure to pace may be misdiagnosed with programmed rate hysteresis. 2Some devices have programmed behavior that may simulate malfunction, termed pseudomalfunction. Primary pacemaker malfunction is rare, accounting for less than 2% of all device-related problems in one large center over a 6-yr period. Pacing malfunction can occur with an implanted pacemaker or ICD because all contemporary ICDs have at least a backup single-chamber pacing capability, and most have dual-chamber pacing as well. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
